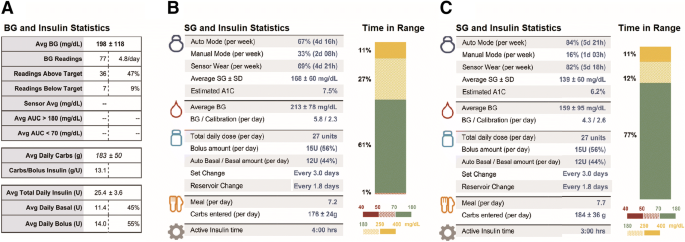
![Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY](https://www.touchendocrinology.com/wp-content/uploads/sites/5/2018/02/table1-summary-of-char.png)
Selecting the Appropriate Continuous Glucose Monitoring System – a Practical Approach | [current-page:pager]touchENDOCRINOLOGY

PDF) Efficacy and safety comparison of continuous glucose monitoring and self-monitoring of blood glucose in type 1 diabetes: Systematic review and meta-analysis

PDF) Prospective multi-center registry to evaluate efficacy and safety of the newly developed diamond-like carbon-coated cobalt-chromium coronary stent system | Atsushi Hirohata - Academia.edu

Reference Guide for Integrating Continuous Glucose Monitoring Into Clinical Practice - Davida F. Kruger, Steve V. Edelman, Deborah A. Hinnen, Christopher G. Parkin, 2019

Insulin Lispro with Continuous Subcutaneous Insulin Infusion is Safe and Effective in Patients With Type 2 Diabetes: A Randomized Crossover Trial of Insulin Lispro Versus Insulin Aspart* - Endocrine Practice

Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf

Continuous Glucose Monitors and Automated Insulin Dosing Systems in the Hospital Consensus Guideline - Rodolfo J. Galindo, Guillermo E. Umpierrez, Robert J. Rushakoff, Ananda Basu, Suzanne Lohnes, James H. Nichols, Elias K.

Medical devices, smart drug delivery, wearables and technology for the treatment of Diabetes Mellitus - ScienceDirect

Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf
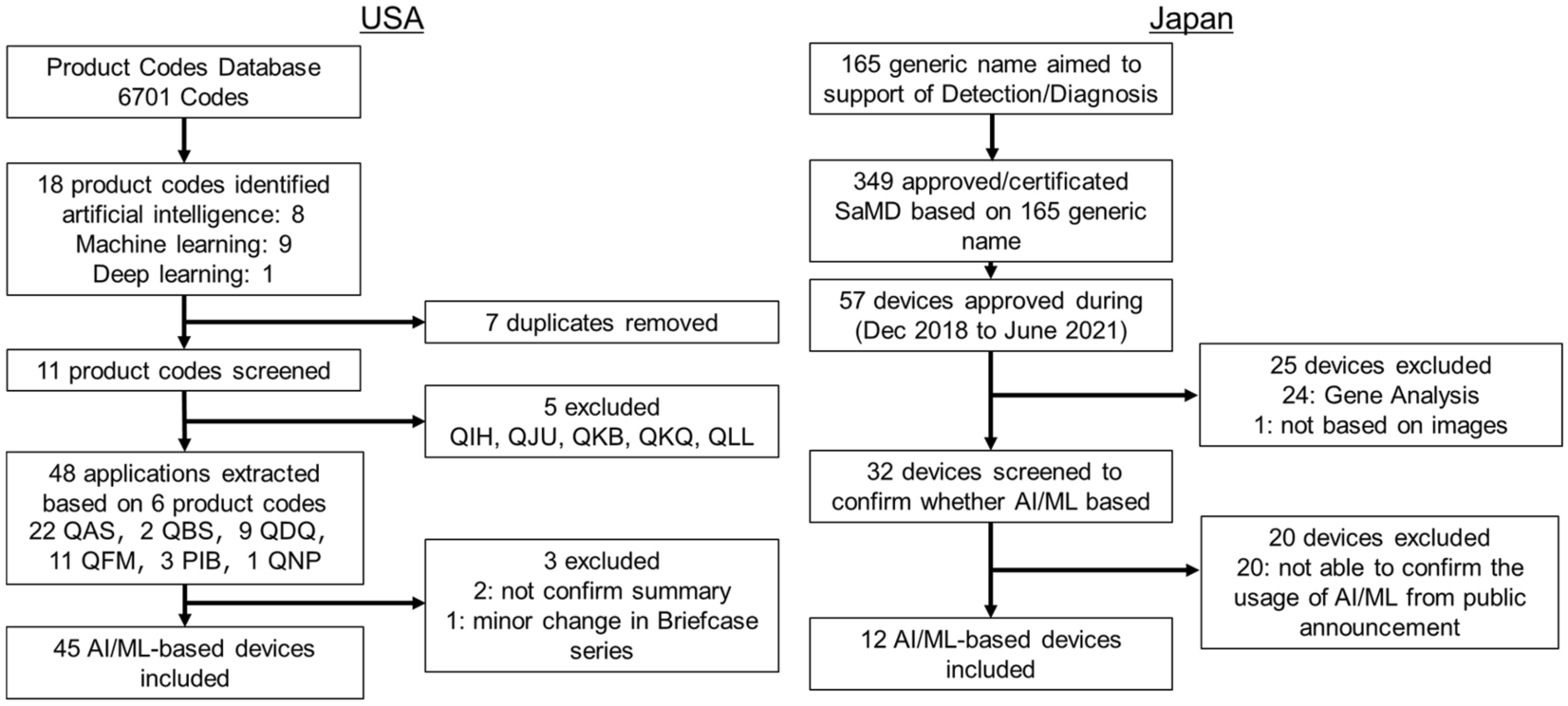
Systematic analysis of the test design and performance of AI/ML-based medical devices approved for triage/detection/diagnosis in the USA and Japan | Scientific Reports

Long-Term Clinical Effectiveness of a Drug-Coated Balloon for the Treatment of Femoropopliteal Lesions | Circulation: Cardiovascular Interventions

Cybersecurity features of digital medical devices: an analysis of FDA product summaries. - Abstract - Europe PMC
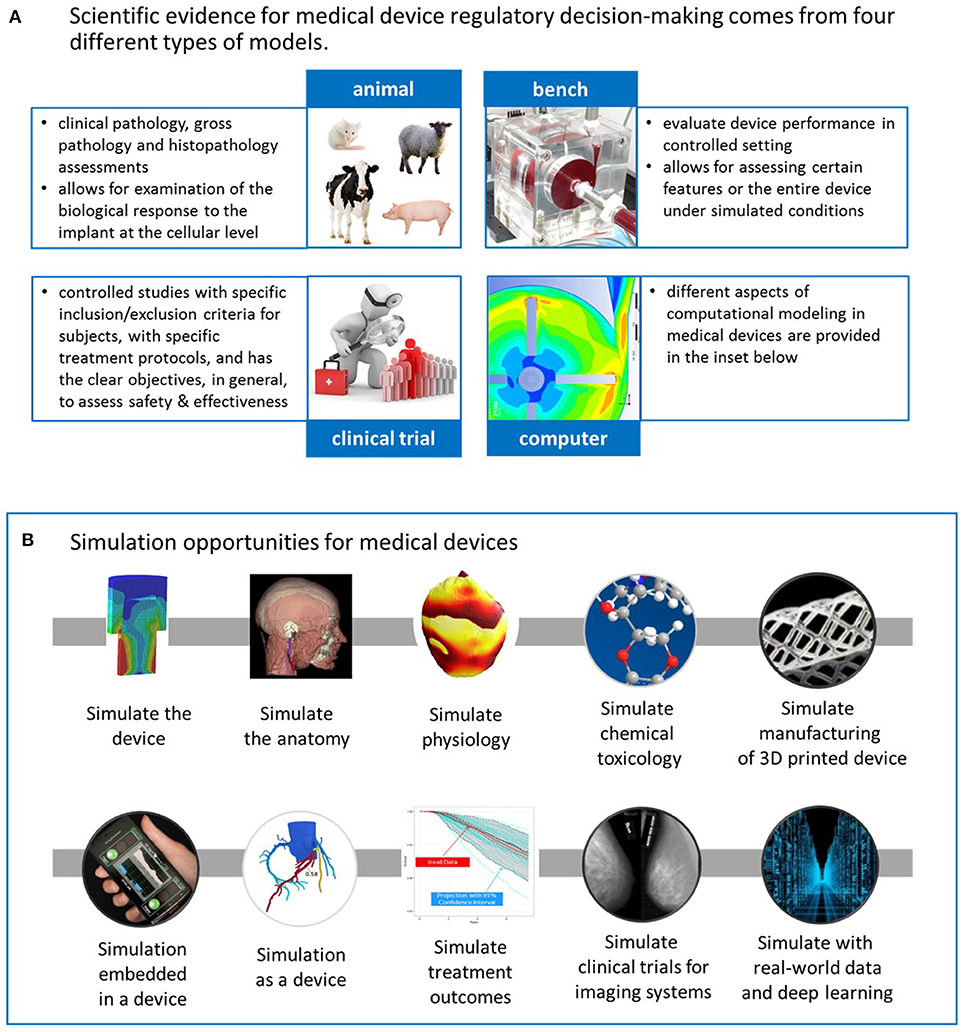
Frontiers | Advancing Regulatory Science With Computational Modeling for Medical Devices at the FDA's Office of Science and Engineering Laboratories
Postmarket Surveillance of Medical Devices: A Comparison of Strategies in the US, EU, Japan, and China | PLOS Medicine

Централно управление Морска анемона грим accessdata.fda minimed summary of safety and effectiveness accessories Насърчаване Парична сума разбирам