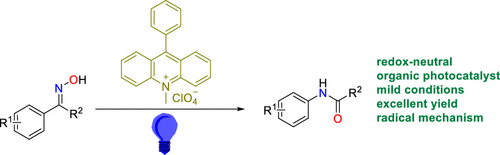
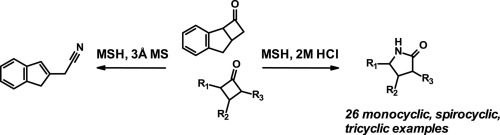
Direct and Stereospecific Synthesis of N-H and N-Alkyl Aziridines from Unactivated Olefins Using Hydroxylamine-O-Sulfonic Acids. - Abstract - Europe PMC
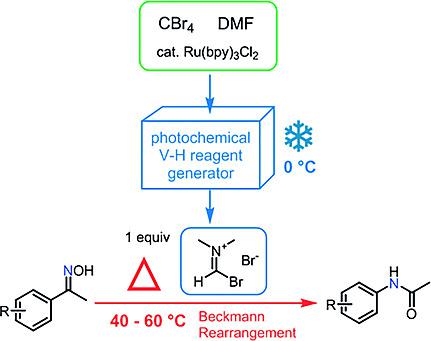
Visible Light‐Promoted Beckmann Rearrangements: Separating Sequential Photochemical and Thermal Phenomena in a Continuous Flow Reactor - Eur. J. Org. Chem. - X-MOL

PDF) The HosA Histone Deacetylase Regulates Aflatoxin Biosynthesis Through Direct Regulation of Aflatoxin Cluster Genes
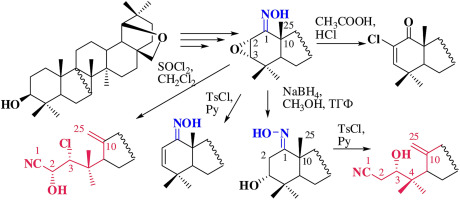
Synthesis of 1,10-seco-triterpenoids by the Beckmann fragmentation from allobetulin,Tetrahedron - X-MOL

Table 1 from Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). | Semantic Scholar

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Abstract - Europe PMC

Cu(OTf)2-catalyzed Beckmann Rearrangement of Ketones Using Hydroxylamine-O-sulfonic Acid (HOSA). - Synthesis - X-MOL
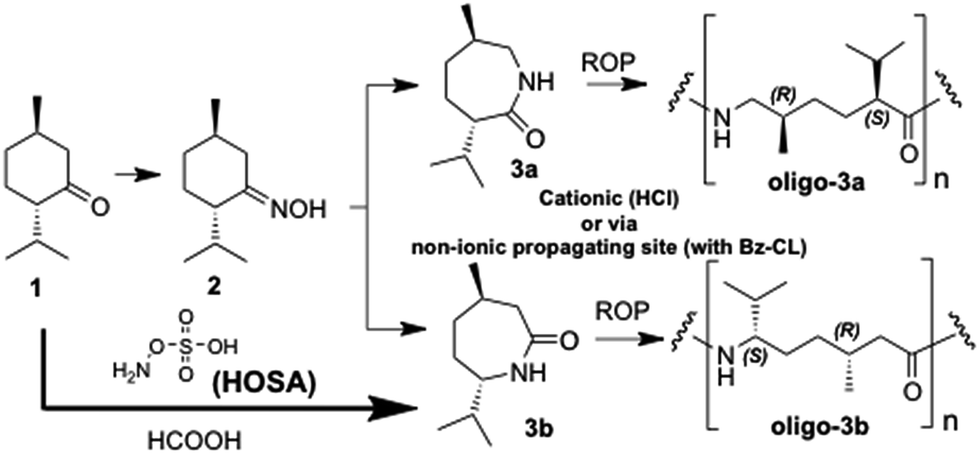
New insights into synthesis and oligomerization of ε-lactams derived from the terpenoid ketone (−)-menthone - RSC Advances (RSC Publishing)
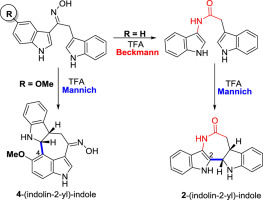
Observations arising from a Beckmann rearrangement-Mannich cyclization approach to the azepinobisindole alkaloid iheyamine A - Tetrahedron - X-MOL