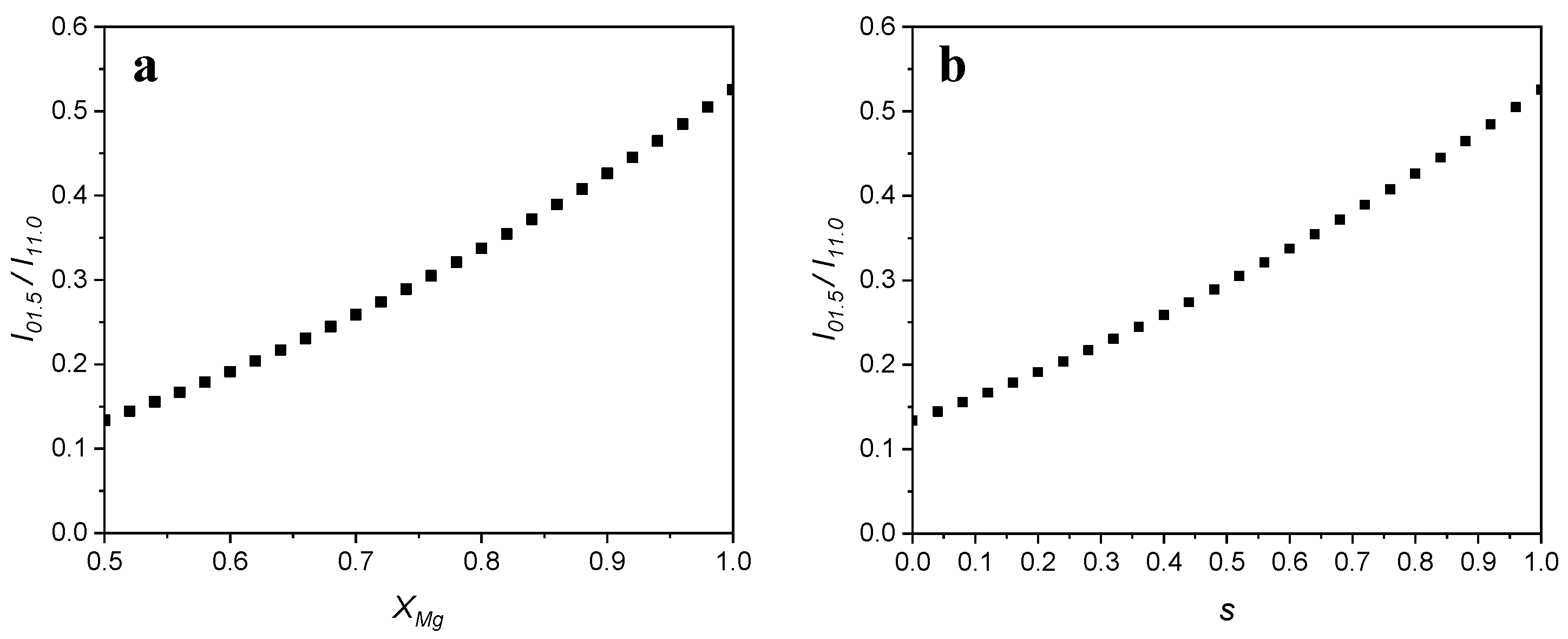
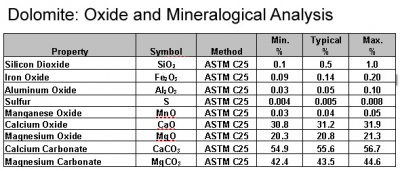
Dissolution profile of dolomite in chloric acid solution: The effect of chloric acid concentration and pulp density

Structure, genesis and resources efficiency of dolomite: New insights and remaining enigmas - ScienceDirect

SOLVED:The mineral dolomite contains magnesium carbonate. This reacts with hydrochloric acid. MgCO3(s)+2 HCl(aq) →CO2(g)+MgCl2(aq)+H2 O(ℓ) (a) Write the net ionic equation for this reaction and identify the spectator ions. (b) What type